×
- Acid
- Acid anhydride
- Atomic Number
- Addition Polymer
- Addition Polymerization
- Alkane
- Alkane:Reactivity
- Alkene
- Alkyne
- Alcohol
- Amphoteric Oxide or Hydroxide
- Alkalis
- Absolute Temperature
- Atom
- Anode
- Anion
- Addition reaction
- Avogadro's number N
- Avogadro's law
- Atomic mass unit (a.m.u)
- Base
- Bond (Chemical)
- Basicity of an acid, monobasic, dibasic, tribasic
- Bronsted-Lowry's definition of acids and bases
- Boyle's law
- Catalyst
- Carbon-12 scale
- Cathode
- Cation
- Charle's law
- Combined gas equation
- Condensation Polymerization
- Compound
- Conductor (electrical)
- Covalent bond
- Coordinate bond
- Coulomb (C)
- Chemical reaction
- Cracking of alkanes
- Deliquescence
- Deposition
- Delocalized electrons
- Diatomic molecule
- Diffusion
- Dilute acid
- Dilution
- Exothermic reaction
- Endothermic reaction
- Equilibrium
- Efflorescent salt
- Enthalpy (ΔH) of reaction
- Electrolyte
- Electrolysis
- Electroplating
- Element
- Empirical formula
- Faraday's first law of electrolysis
- Formula equation
- Formula unit and formula mass
- Functional group
- Gay-Lussac's law
- Graham's law
- Half-life of radioactive decay
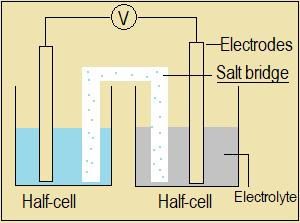
- Half-cell
- Halogenation
- Hard water
- Hardening of vegetable oil
- Hydrogen
- Hydrogen bond
- Hydrated salt
- Hydrocarbon
- Hydrogen ion as a proton
- Hygroscopic salt
- Ion
- Ionic Discharge
- Ionic or electrovalent bond
- Ionic migration
- Ionization energy
- Isotopes
- Le Chartelier's principle
- Markownikoff's rule
- Material
- Mineral
- Mixture
- Mole
- Molarity
- Molecule
- Neutralization
- Nuclear fission
- Relative Atomic Mass
- Radioactive decay
- Reduction
- Reduction half-cell
- Redox processes
- Radioactive Isotope
- Radioactivity
- Salt bridge
- Substitution reaction
- Solvent
- Solute
- Solution
- Saturated solution
- Salt
- Solubility
- Soft water
- Structural formula
- Suspension
- Sublimation
- Substance:Chemical substance
- Strong acid
Salt bridge
This is a strip of absorbent paper soaked in a salt solution and
used to complete a circuit between two half-cells.
Examples: A strip of filter paper soaked in potassium nitrate
solution or sodium nitrate solution
OTHER TOPICS (FULL)

INTRODUCTION TO CHEMISTRY
SIMPLE CLASSIFICATION OF SUBSTANCES AND SEPARATION OF MIXTURES
ACIDS, BASES AND INDICATORS

AIR AND COMBUSTION

WATER AND HYDROGEN
STRUCTURE OF THE ATOM, AND THE PERIODIC TABLE

CHEMICAL FAMILIES AND PATTERNS IN PROPERTIES

CHEMICAL BONDING AND STRUCTURE

SALTS

EFFECT OF AN ELECTRIC CURRENT ON SUBSTANCES
CARBON AND SOME OF ITS COMPOUNDS
