CHEMISTRY FORM 2
- 1.1 Structure of the atom
- 1.2 Atomic Number and Mass Number
- 1.3 Isotopes
- 1.4 Energy levels and electron arrangement
- 1.5 Development of the Periodic Table
- 1.6 Relative Atomic Mass and Isotopes
- 1.7 Ion Formation
- 1.8 Chemical Formulae
- 1.9 Chemical Equations

- 2.1 Alkali metals (Group I elements)
- 2.2 Alkali Earth Metals (Group II elements)
- 2.3 Halogens (Group VII elements)
- 2.4 Noble gases (Group VIII elements)
- 2.5 Properties and Trends Across the Periodic Table

- 3.1 Bond
- 3.2 Ionic bond
- 3.3 Giant ionic structure
- 3.4 Covalent bond
- 3.5 Co-ordinate bond
- 3.6 Molecular structures
- 3.7 Giant covalent structures
- 3.8 Metallic Bond
- 3.9 Types of bond across a period
- 3.10 Oxides of elements in Period 3
- 3.11 Chlorides of Period 3 elements

- 4.1 What is a salt?
- 4.2 Types of salt
- 4.3 Solubility of salts in water
- 4.4 Methods of preparing salts
- 4.4.1 Reacting a Metal with an Acid
- 4.4.2 Reacting an Acid with a Base (Neutralization)
- 4.4.3 Reacting an Acid with a Carbonate (or hydrogencarbonate of metal)
- 4.4.4 Combining elements Directly (Direct Combination of elements)
- 4.4.5 Precipitation (Double decomposition)
- 4.5 Action of heat on salts
- 4.6 Uses of salts

- 5.1 Electrical conduction
- 5.2 Electrical conductivity of molten substances
- 5.3 Electrical conductivity of substances in aqueous state
- 5.4 Electrolysis
- 5.5 Applications of electrolysis
- 6.1 Allotropes of carbon
- 6.2 Chemical properties of carbon
- 6.3 Carbon (IV) oxide
- 6.4 Carbon (II) oxide (CO)
- 6.5 Large scale production of sodium carbonate and sodium hydrogencarbonate
- 6.6 Effect of carbon (II) oxide and carbon (IV) oxide on the environment
- 6.7 Carbon cycle

Salts: Combining elements Directly (Direct Combination of elements)
4.0 Salts
4.4.4 Combining elements Directly (Direct Combination of elements)
In Section 2.1 of Topic 2, we saw that alkali metals burn in chlorine gas. Open the video showing the burning of alkali metals in chlorine gas to observe the reactions once more.
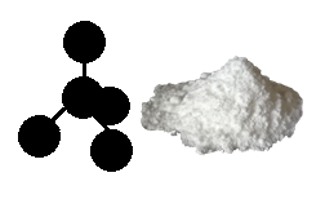
These are typical examples of direct combination reactions. Figure 4.4.4 shows sodium chloride produced by direct combination of sodium and chlorine. The salts produced by this method look powdery (very fine crystals).

Figure 4.4.4(a) Sodium chloride salt
The reaction between iron and sulphur used earlier to demonstrate chemical change in Volume 1, is another example of direct combination of elements. Where both elements are solids, they should be in powder form, mixed uniformly then heated strongly in a test tube till the glow stops.
For a metal and gas, the gas is first prepared, dried, then passed over the metal heated strongly in a combustion tube.
Questions 4.4.4
- Draw a diagram of the set-up you would use to combine (react) dry chlorine gas with iron metal to form iron (III) chloride.
- Why should the gas be dry?
- How can the gas be dried?
- Write a chemical equation for direct combination of iron and chlorine gas.
- Why is strong heating necessary?
- How is chlorine gas produced?
Answers to Questions 4.4.4

Figure 4.4.4(b) Anhydrous iron (III) chloride
