×








CHEMISTRY LEVEL 3
1. GAS LAWS
- 1.1 Boyle's Law
- 1.2 Charles'law
- 1.3 Combined gas law
- 1.4 Standard conditions
- 1.5 Diffusion and Graham's law
2. THE MOLE: Formulae and Chemical Equations
- 2.1 Relative Mass
- 2.2 Atoms, Molecules and Moles
- 2.3 Compounds and the mole
- 2.4 Empirical and Molecular formula
- 2.5 Concentration of a solution
- 2.6 Molar solutions
- 2.7 Preparation of molar solutions
- 2.8 Dilution of a solution
- 2.9 Stoichiometry of chemical reactions
- 2.10 Volumetric analysis
- 2.11 Titration
- 2.12 Redox titration
- 2.13 Atomicity and molar gas volume
- 2.14 Combining volumes of gases
3. ORGANIC CHEMISTRY 1
- 3.1 Alkanes
- 3.1.1 Formulae of alkanes
- 3.1.2 Cracking of alkanes
- 3.1.3 Nomenclature (systematic naming) of alkanes
- 3.1.4 Isomerism in alkanes
- 3.1.5 Laboratory preparation of alkanes
- 3.1.6 Physical properties of alkanes
- 3.1.7 Chemical properties of alkanes
- 3.1.8 Uses of alkanes
- 3.2 Alkenes
- 3.2.1 Nomenclature of alkenes
- 3.2.2 Isomerism in alkenes
- 3.2.3 Laboratory preparation of ethene
- 3.2.4 Physical properties of alkenes
- 3.2.5 Chemical properties of alkenes
- 3.2.6 Test for alkenes
- 3.2.7 Uses of alkenes
- 3.3 Alkynes
- 3.3.1 Nomenclature of alkynes
- 3.3.2 Isomerism in alkynes
- 3.3.3 Laboratory preparation of ethyne
- 3.3.4 Physical properties of alkynes
- 3.3.5 Chemical properties of alkynes
- 3.3.6 Test for alkynes
- 3.3.7 Uses of alkynes
- 3.4 Recommended practice of topic summary
4. NITROGEN AND ITS COMPOUNDS
- 4.1 Extraction of nitrogen from air
- 4.2.1 Laboratory preparation of nitrogen gas from the air
- 4.2.2 Laboratory preparation of nitrogen gas from ammonium nitrite ((NH4NO2))
- 4.2.3 Uses of nitrogen
- 4.3 Oxides of nitrogen
- 4.3.1 Nitrogen (I) oxide
- 4.3.2 Nitrogen (II) oxide
- 4.3.3 Nitrogen (IV) oxide
- 4.4.1 Laboratory preparation of ammonia
- 4.4.2 Solubility of ammonia in water
- 4.4.3 Reactions of aqueous ammonia (ammonia solution)
- 4.4.4 Reactions of ammonia gas
- 4.4.5 Industrial manufacture of ammonia: The Haber Process
- 4.4.6 Uses of ammonia
- 4.4.7 Nitrogenous fertilizers
- 4.5.1 Laboratory preparation of nitric (V) acid
- 4.5.2 Industrial manufacture of nitric (V) acid
- 4.5.3 Reactions of dilute nitric (V) acid
- 4.5.4 Reactions of concentrated nitric (V) acid
- 4.5.5 Uses of nitric (V) acid
- 4.6.1 Action of heat on nitrates
- 4.6.2 Test for nitrates (nitrate ions, NO3-)
- 4.6.3 Air pollution by nitrogen compounds
- 4.7 Summary on nitrogen and its compounds
5. SULPHUR AND ITS COMPOUNDS
- 5.0 Sulphur and its Compounds
- 5.1.1 Extraction of sulphur
- 5.1.2 Allotropes of sulphur
- 5.1.3 Physical properties of sulphur
- 5.1.4 Chemical properties of sulphur
- 5.2.1 Preparation of sulphur (IV) oxide
- 5.2.2 Physical properties of sulphur (IV) oxide
- 5.2.3 Chemical properties of sulphur (IV) oxide
- 5.2.4 Reducing action of sulphur (IV) oxide
- 5.2.5 Oxidization of SO2 to SO3
- 5.2.6 Oxidizing action of sulphur (IV) oxide
- 5.2.7 Test for sulphite (SO32-) and sulphate (SO42-) ions
- 5.2.8 Uses of sulphur (IV) oxide
- 5.3 Large scale (industrial) manufacture of sulphuric (VI) acid
- 5.3.1 Physical properties of concentrated sulphuric (VI) acid
- 5.3.2 Chemical properties of concentrated sulphuric (VI) acid
- 5.3.3 Reactions of dilute sulphuric (VI) acid
- 5.4 Hydrogen sulphide
- 5.4.1 Chemical properties of hydrogen sulphide
- 5.4.2 Air pollution by compounds of sulphur
- 5.5 Summary on sulphur and its compounds
6. CHLORINE AND ITS COMPOUNDS
- 6.1 Occurrence of chlorine
- 6.2 Laboratory preparation of chlorine
- 6.3 Physical properties of chlorine
- 6.4 Chemical properties of chlorine
- 6.5 Oxidizing properties of chlorine
- 6.6 Reaction of chlorine with alkaline solutions
- 6.7 Test for chloride ions
- 6.8 Uses of chlorine and its compounds
- 6.9 Preparation of hydrogen chloride gas
- 6.10 Physical properties of hydrogen chloride
- 6.11 Chemical properties of hydrogen chloride
- 6.12 Industrial manufacture of hydrochloric acid
- 6.13 Uses of hydrochloric acid
7. A guide to chemical tests based on this module

Content developer

Nitrogen and its Compounds: Action of heat on nitrates
4.0 Nitrogen and its Compounds
4.6 Nitrates
4.6.1 Action of heat on nitrates
Watch the video demonstrations of the action of heat on the sampled nitrate salts, alongside Figure 4.6.1 and record your observations. Note in particular, the colour, sound, and any effects on glowing splint and wet litmus paper.
(courtesy Youtube-Chemistry SPM: Learn 6.10 Effect of Heat on Nitrate Salts In 4 Minutes by ChemTube)
| Metal nitrate | Changes during heating of the nitrate |
|---|---|
| Sodium nitrate (NaNO3) | 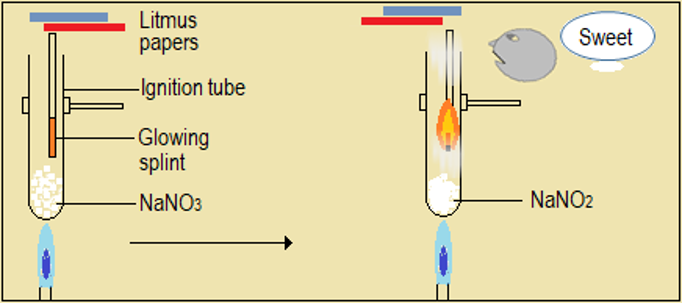
|
| Potassium nitrate (KNO3) | 
|
| Ammonium nitrate (NH4NO3) | 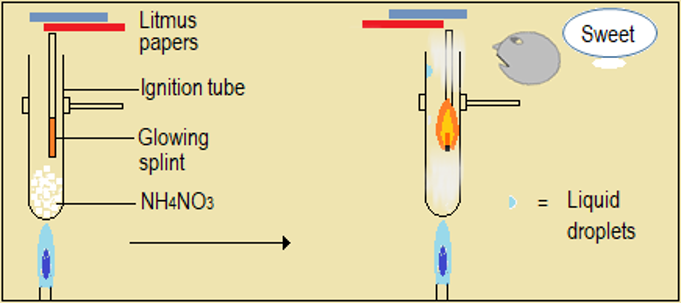
|
| Copper (II) nitrate (Cu(NO3)2) | 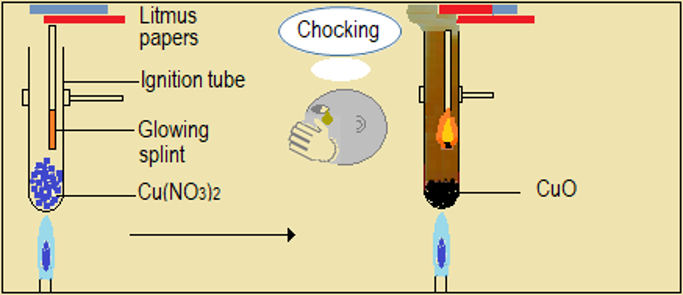
|
| Zinc nitrate (Zn(NO3)2) | 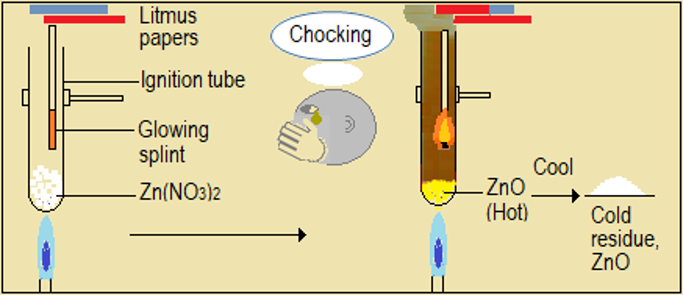
|
| Lead (II) nitrate (Pb(NO3)2) | 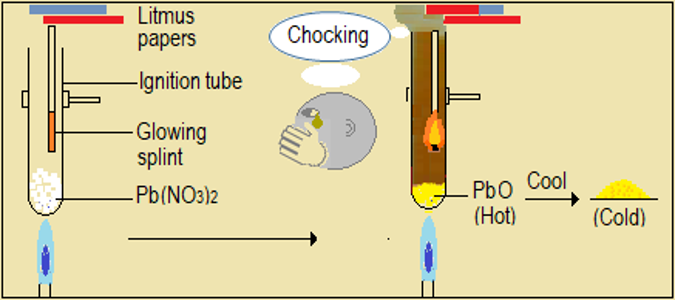
|
Figure 4.6.1: Effect of heat on nitrates
Questions 4.6.1
- State and explain the common observations when nitrates are heated.
- Which observations apply to nitrates of heavy metals (e.g. copper, zinc, and lead) only?
- How does the observation with ammonium nitrate differ from that of other nitrates?
- Write equations for thermal decomposition of each of the following nitrates.
- Sodium nitrate (NaNO3)
- Potassium nitrate (KNO3)
- Ammonium nitrate (NH4NO3)
- Copper (II) nitrate (Cu(NO3)2)
- Zinc nitrate (Zn(NO3)2)
- Lead (II) nitrate (Pb(NO3)2)
- Silver nitrate (AgNO3) NB: Hot silver oxide is unstable and readily decomposes to silver metal and oxygen gas.
- Mercury (II) nitrate (Hg(NO3)2)
- While heating a salt, when should we begin to suspect that it is a nitrate?
Answers to Questions 4.6.1 
